Flawless Balance Quality assurance program
Product quality assurance and logistics system that best assures the delivery of a quality product to the customer.
FSVP...... foreign suppliers .....
- The decision to bring a new product to market is made based on market demands and our potential ability to add quality and diversification to the market segment
- We request the certification of compliance from out Manufacturing partner, and keep these registrations and inspection reports on file "herein"
- We evaluate the product specifications as well as ensure the levels of toxic metals and or allergens are well below required limits of the, (FDA) food and drug administration.
- We then order samples and begin LD 50 animal testing to help to ensure safety of our customers. We contract with a third party company to have this test completed.
- We continually ask our Manafacturer for certifications from outside independent firms that check and recheck our Manafacturer's quality programs. "See attached certificates" we then ask for the latest inspection certificates for our records
- Once the samples, certificates of analysis and inspection certificates are reviewed and we receive positive news regarding the LD50 test we order a sample Manufacturing initial run normally under 1000 units
- We design our label according to the FDA guidelines, our company lists the ingredients, disclaimers such as " this product has not been evaluated by the FDA and is not intended to diagnose treat prevent delay or cure any disease". We plan on a using a conservative marketing and labeling approach with our products. Our goal is to provide a benefit without overselling our products.
- To assure lot control we stamp the Manufacturing and expiration date on each bottle. And on label on each the edge of each bag packaged product.
- Stamped Dates are completed at the factory before the product is shipped. This process was made effective Dec 2017. Between march 2015 and December 2017 we used a different serial number that includes the Manufacturing date for each shipment, now we do both for the expiration date is and always has been coded into the labels barcode.
- As of 2018 will introduced our new logo and a gloss label for our new product Flawless Balance. We will also change our bottle to a uv protective blue bottle with a two layer tear away plastic Tampere resistant cap, along with an inner seal that must be broken before usage. This double seal prevents the possibility of product tampering for safety.
- Most of our Sales are internet based we evaluate our clients feedback and aways do what is possible to improve our customers satisfaction levels.
- Shipping and logistics. Since most of our sales are internet based our shipping labels are derived from this information. Products are shipped primarily from our New Jersey warehouse or by our sales person / owner operator in New Jersey.
- If a product delivery is delayed tracking numbers are checked and or sent to the customer
- Our inventory is primarily housed in our pennsuaken New Jersey warehouse with a small amount stored in our west Coast Warehouse in California.
- Sales and marketing is primarily internet and trade show based. With some owner operator direct sales promotion.
- We have recently began adding sales representative whom are all trained not to make medical claims and to adhere to companies policies listed on the footer of each of our web pages.
Product Name Formula 1 Manufacture Date 2017/11/10
Latin Name Report Date 2017/11/17
Batch Quantity 750mg Sampling Date 2017/11/14
Batch Number HK171110 Expiration Date 2020/11/09
Composition Specification Results Methods
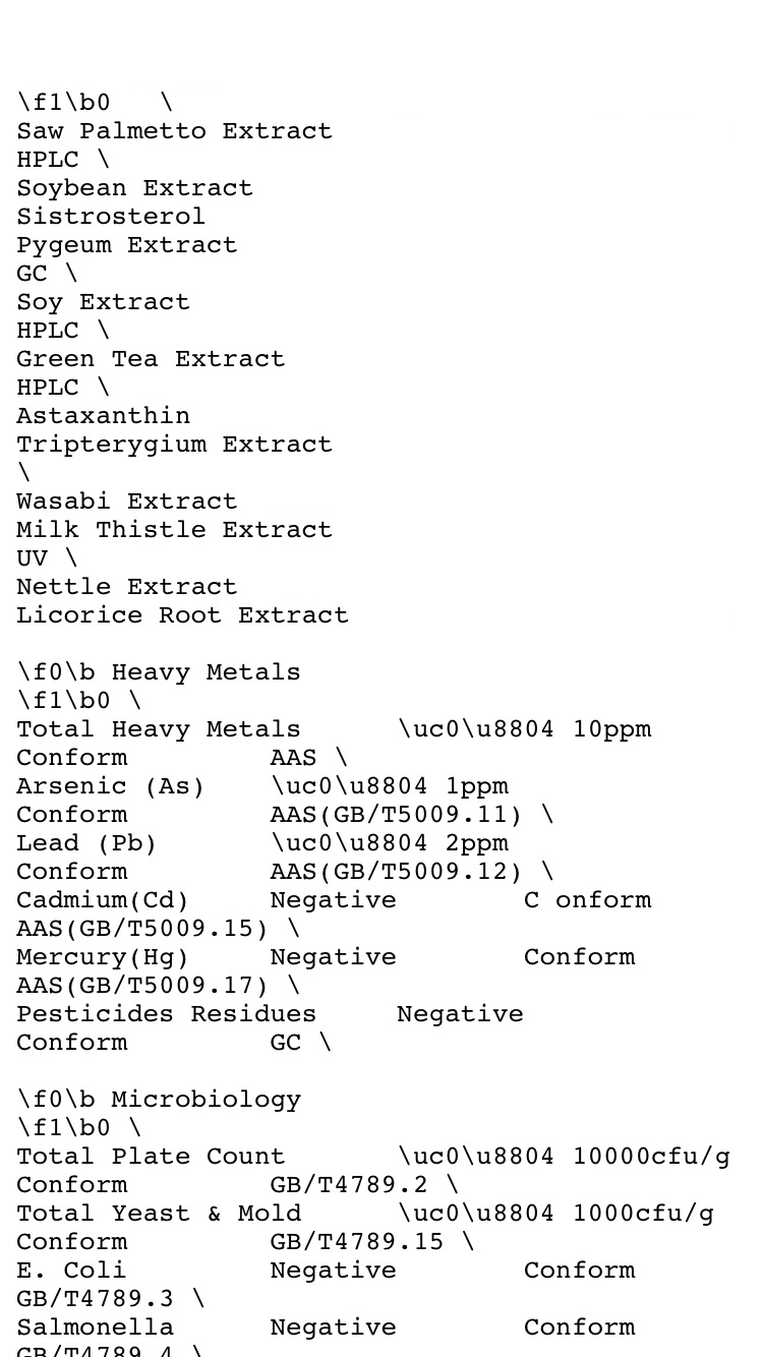
Saw Palmetto Extract
Soybean Extract
Pygeum Extract
Soy Extract
Green Tea Extract
Astaxanthin
Tripterygium Extract
Wasabi Extract
Milk Thistle Extract
Nettle Extract
Licorice Root Extract
Heavy Metals
Total Heavy Metals ≤10ppm Conform AAS
Arsenic (As) ≤1ppm Conform AAS(GB/T5009.11)
Lead (Pb) ≤2ppm Conform AAS(GB/T5009.12)
Cadmium(Cd) Negative C onform AAS(GB/T5009.15)
Mercury(Hg) Negative Conform AAS(GB/T5009.17)
Pesticides Residues Negative Conform GC
Microbiology
Total Plate Count ≤10000cfu/g Conform GB/T4789.2
Total Yeast & Mold ≤1000cfu/g Conform GB/T4789.15
E. Coli Negative Conform GB/T4789.3
Salmonella Negative Conform GB/T4789.4
Staphylococcus Negative Conform GB/T4789.10
Packing and Storage Ambient temperature range from 18 to 26 degrees C(46.4 to 78.8 Degree F), humidity 50%. Protected from strong light and heat. Typicallly inside 25kg or client’s requirement, with food grade plastic bags, out side Paper -Drums
Shelf Life 3 Year When Stored properly
Expiration Date 3 Year
Latin Name Report Date 2017/11/17
Batch Quantity 750mg Sampling Date 2017/11/14
Batch Number HK171110 Expiration Date 2020/11/09
Composition Specification Results Methods
Saw Palmetto Extract
Soybean Extract
Pygeum Extract
Soy Extract
Green Tea Extract
Astaxanthin
Tripterygium Extract
Wasabi Extract
Milk Thistle Extract
Nettle Extract
Licorice Root Extract
Heavy Metals
Total Heavy Metals ≤10ppm Conform AAS
Arsenic (As) ≤1ppm Conform AAS(GB/T5009.11)
Lead (Pb) ≤2ppm Conform AAS(GB/T5009.12)
Cadmium(Cd) Negative C onform AAS(GB/T5009.15)
Mercury(Hg) Negative Conform AAS(GB/T5009.17)
Pesticides Residues Negative Conform GC
Microbiology
Total Plate Count ≤10000cfu/g Conform GB/T4789.2
Total Yeast & Mold ≤1000cfu/g Conform GB/T4789.15
E. Coli Negative Conform GB/T4789.3
Salmonella Negative Conform GB/T4789.4
Staphylococcus Negative Conform GB/T4789.10
Packing and Storage Ambient temperature range from 18 to 26 degrees C(46.4 to 78.8 Degree F), humidity 50%. Protected from strong light and heat. Typicallly inside 25kg or client’s requirement, with food grade plastic bags, out side Paper -Drums
Shelf Life 3 Year When Stored properly
Expiration Date 3 Year